The Dutch Pulmonary Hypertension Association, Stichting Pulmonale Hypertensie, has contributed financially with an amount of € 40.000 the RECOMPENSE – Right VEntricular COMPENsation with SotatercEpt – Phase 4 study conducted by the Amsterdam UMC. This research aims to investigate sotatercept’s effects on right ventricular function.
Background
Several large international studies have already been conducted in which patients with pulmonary arterial hypertension were treated with sotatercept, says Eszter Tóth, who is conducting the study together with Prof. Dr. Harm Jan Bogaard and Prof. Dr. Frances de Man. These studies have shown that there is improvement following treatment with sotatercept. Pulmonary artery pressure decreases, and exercise tolerance improves. “Now that the drug has been approved for commercial use in the European Union, it may be used alongside current treatments for pulmonary arterial hypertension in patients who still experience functional limitations”, she explains. “However, since the Dutch Health Care Institute still needs to decide whether sotatercept will be included in the reimbursement system, patients currently only have access to it within research settings.”
How does Sotatercept work?
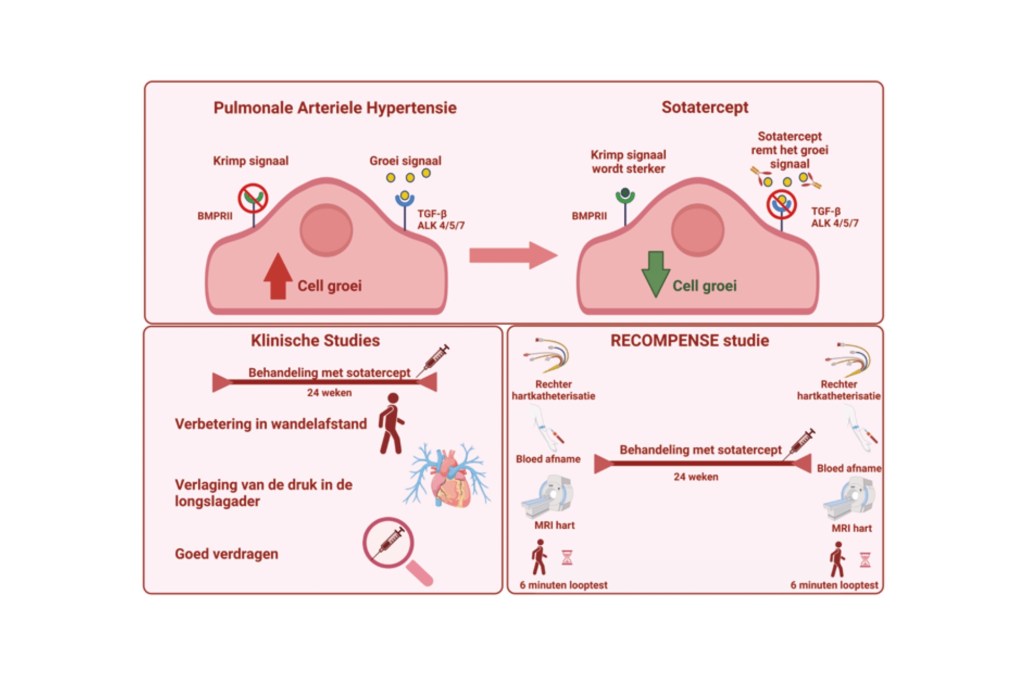
Sotatercept targets the BMP/TGF-β signaling pathway, Eszter explains. “This means the drug affects how signals are transmitted within cells. Current pulmonary arterial hypertension treatments focus on dilating blood vessels. Sotatercept, on the other hand, approaches the disease process differently by reversing vascular remodeling. This means that the process of the blood vessel walls thickening is slowed down or stopped.”
Why conduct a new study?
The positive results of sotatercept suggest that the reduction in pulmonary artery pressure also decreases the strain on the right ventricle of the heart, potentially restoring its function, Eszter explains. “Current therapies do not reduce the burden on the right ventricle. This is a potential long-term benefit of sotatercept. However, at present, we lack data on the effects of sotatercept on the heart. Previous studies have primarily focused on the clinical effects of sotatercept and pulmonary artery pressure. Our research aims to investigate sotatercept’s effects on right ventricular function.”
Study Design
The study, called RECOMPENSE, is a prospective, single-arm, open-label study. Single-arm, open-label means there is no placebo, and all patients included in the study receive sotatercept. Prospective means that participants are followed over time—in this case, for up to 30 weeks. Before enrolling in the study, participants undergo a screening, Eszter explains. “All candidates attend a screening visit to check whether they meet the study’s inclusion criteria.” This includes a physical examination, blood tests, a six-minute walk test, right heart catheterization (RHC), and cardiac magnetic resonance imaging (cMRI) with contrast agent.
All enrolled patients then receive subcutaneous injections of sotatercept, starting with a dose of 0.3 mg/kg. Patients visit the hospital every three weeks for blood tests, physical examinations, sotatercept administration, and evaluation of any side effects. If blood parameters (specifically hemoglobin and platelet levels) remain stable after the initial dose, the dosage is increased to 0.7 mg/kg. This 0.7 mg/kg dose is the maximum dosage in this study. After 24 weeks of sotatercept therapy, there is an end-of-study visit, including a physical examination, a six-minute walk test, right heart catheterization, and cMRI with contrast agent.
Eligibility Criteria
Adult patients with heritable or idiopathic PAH—with mPAP > 20 mmHg, PCWP or LVEDP ≤ 15 mmHg, and PVR ≥ 4WU as measured by right heart catheterization—are eligible for the study. Participants must be stable on their current pulmonary arterial hypertension therapies. Patients with a history of bleeding disorders or low blood pressure, or who have recently been hospitalized due to pulmonary arterial hypertension, are excluded. Additionally, NT-proBNP levels must be above 300 ng/L during the screening visit. Female participants must use contraception and undergo regular pregnancy tests to participate.
What does participation mean for patients?
Participation involves frequent hospital visits and blood tests, which can be physically and emotionally taxing, Eszter explains. “However, these steps are essential to ensure the safe monitoring and administration of sotatercept.” Participants also undergo invasive diagnostics such as right heart catheterization and cMRI with contrast agent during the baseline and end-of-treatment visits. “These procedures are necessary because they provide the most accurate measurements of pulmonary pressures, which cannot be obtained using less invasive methods.” The cMRI, enhanced with gadolinium contrast, allows for the assessment of cardiac fibrosis. “Although gadolinium is generally considered safe, allergic reactions and mild side effects such as headache, dizziness, nausea, or localized hot or cold sensations at the injection site may occur,” Eszter adds.
Potential Side Effects of Sotatercept
Sotatercept is generally well tolerated, Eszter notes. Known side effects include elevated hemoglobin levels, low platelet counts, telangiectasia (small dilated blood vessels causing a red appearance), headache, diarrhea, and peripheral edema. “The long-term safety and rare potential side effects remain unknown. This poses a risk for patients. However, Phase 2 and Phase 3 studies concluded that sotatercept is safe for pulmonary arterial hypertension patients and that its benefits outweigh the risk profile.”
The Importance of the Dutch PH Association’s Contribution
“The financial support from the Dutch Pulmonary Hypertension Association makes it possible for us to conduct this study,” says Eszter. “In addition to the clinical research, we will use the contribution to better understand how sotatercept works at the cellular level. At Amsterdam UMC, we can recreate the heart in the lab in 3D using patient-derived blood cells. These can be genetically modified into cardiomyocytes, inflammatory cells, endothelial cells, and fibroblasts. This allows us to study the effects of sotatercept on the heart at the cellular level and work toward the development of new medications with fewer side effects.”
Click on this link for more information about the study.
Text: Anita Harte