Download the webinar slides here
Learning objectives of the webinar
Our goal is to empower individuals living with pulmonary hypertension and their families, equipping them with actionable insights and practical knowledge.
- Understand the current PROM tools and how they were developed.
- Recognize why PROMs should be included in routine patient care, particularly at PH centers.
- Explore the potential use of PROMs in clinical trials, including incorporating the patient perspective in trial design.
- Discuss what is needed to improve current PROM tools and develop future ones.
Transcript
NB. This transcript can be translated into your preferred language – use orange button at the bottom centre of this page to select it (40 languages available)
DISCLAIMER: Despite every effort to ensure the accuracy of this transcript, we strongly encourage all visitors to consult with their healthcare professionals before making any decisions based on the information provided. Additionally, while the quality of Google Translate has improved tremendously in recent years, please remember that it is an automated service and not a human translation.
DR. ANDREAS REIMANN
Good afternoon or good morning, depending on your location. I’m Andreas Reimann, co-founder and managing partner at admedicum, and I’m delighted to welcome you to today’s event. We’re proud to support the Alliance for Pulmonary Hypertension, a patient-led nonprofit initiative focused on sharing knowledge about pulmonary hypertension. One of the most exciting developments we’ll be discussing today is the joint guidelines of the European Respiratory Society and the European Society of Cardiology, published last September. Our previous webinar centered on shared decision-making, and I’m thrilled to have Dr. Simon Gibbs and Gergely Meszaros joining us today to delve into another crucial aspect of these guidelines: the application of patient-reported outcome measures. Throughout the session, please feel free to ask questions in the chat, which will be monitored by my colleague Robert Pleticha and myself. Following the joint presentation by Dr. Gibbs and Mr. Meszaros, we’ll have an opportunity for discussion and further exploration of their insights. Without further ado, I’m pleased to hand over to Dr. Gibbs.
DR. SIMON GIBBS
Thank you, Andreas, and greetings to everyone joining this webinar globally. Before we dive in, I’d like to disclose my conflicts of interest. While Gergely has none, I’m involved with various pharmaceutical companies in clinical trials and have contributed to the development of one of the patient-reported outcome measures we’ll discuss. Now, let’s move on to our topic of the day: patient-reported outcome measures.

Patient-reported outcome measures originated in mental health research around the 1960s and have since expanded to other medical fields. They can be generic or disease-specific, focusing on symptoms, impact on life, and quality of life. Speaking of quality of life, it encompasses various aspects of a person’s perception, including physical health, psychological state, independence, social

relationships, and environmental factors. The World Health Organization (WHO) defines it as an individual’s perception of their life situation. Incorporating quality of life assessments in patient care, as recognized by the World Health Organization, can enhance healthcare outcomes.

Now, onto the guidelines. The European Respiratory Society (ERS) and the European Society of Cardiology (ESC) jointly released guidelines last year, emphasizing the importance of World Health Organization in patient care. Gergely, perhaps you can elaborate on the European Respiratory Society and European Society of Cardiology guidelines and highlight the key points from the new guidelines regarding PROMs.
GERGELY MESZAROS
Thank you, Simon, and a warm welcome to all attendees joining this webinar from across the globe. It’s truly a pleasure to have this joint session with Professor Simon Gibbs. As Simon mentioned, we often use abbreviations like “ESC” for the European Society of Cardiology and “ERS” for the European Respiratory Society. These two associations have a longstanding history of collaboration, particularly in issuing guidelines on pulmonary hypertension. While the concept of quality of life is not new, its inclusion in the guidelines represents a significant advancement. In the previous guidelines from 2015, there were references to quality of life, but no specific section dedicated to it or patient-reported outcome measures.

In the latest guidelines, however, we felt it was practical to have a dedicated section to quality of life. We relied on definitions from both the US Food and Drug association (FDA) and European Medicines Agency (EMA), emphasizing the key element of self-reporting, as patient reported outcome measures originate directly from the patient. Despite limited characters in scientific papers, we expanded on this definition, focusing on the patient’s experience living with pulmonary hypertension and its impact on both the patient and any caregivers. We outlined various domains, including intellectual, psychosocial, and goal-oriented dimensions. The guidelines provide different levels of recommendation, with green indicating a strong recommendation, and A1 representing the highest level. “Should” and “might be considered” may seem similar, but the former carries more weight in terms of evidence. Unfortunately, due to insufficient evidence, we couldn’t recommend the routine use of quality-of-life measures in clinical practice. While this may seem like bad news, it actually presents an opportunity for improvement. We need to gather more evidence and explore how to integrate quality of life questionnaires into daily patient care effectively. Quality of life is not a standalone section; rather, it’s intertwined with various aspects of patient management.

Let’s continue to explore this further in our discussion. Could we please proceed to the next slide, Andreas? Thank you.
DR. ANDREAS REIMANN
Yes, I apologize for the interruption. Could we please revisit the previous slide to ensure clarity for everyone in the audience? Thank you. Regarding the orange boxes displayed, am I correct in understanding that they signify options that may be considered? To clarify, they neither come with a direct recommendation nor advise against their use; they simply indicate possibilities open for consideration, is that correct?
GERGELY MESZAROS
Yes, that is correct. I understand there might have been some confusion earlier, and I appreciate the opportunity to clarify further. The green indicator represents recommendations supported by substantial clinical data and scientific research. On the other hand, the orange indicator, while not as strongly recommended as the green, does not imply a recommendation against its use. Instead, we recognize its potential value, particularly in enhancing patient care. It represents a beneficial option worth considering, even though the supporting evidence may not be as extensive.”
DR. ANDREAS REIMANN
Thank you very much
GERGELY MESZAROS
I believe that until the next set of guidelines are published, we should endeavor to gather real-world data and additional evidence. This effort will enable us to reevaluate and potentially elevate the status of certain options from orange to green.
DR. ANDREAS REIMANN
Thanks very much Gergely for pointing this out.
GERGELY MESZAROS
Thank you for your question. As I mentioned earlier, quality of life questionnaires and patient-reported outcome measures are not isolated tools. They are integrated not only within the guidelines but also in everyday patient care.
In the context of a multidisciplinary team approach, it is crucial that patients actively contribute data regarding their condition. This input is essential both in discussions and decision-making processes about their care. We firmly believe, from a patient perspective, that this involvement is vital, and patient-reported outcome measures serve as the primary tool for facilitating such contributions. When we discuss multidisciplinary teams and patient empowerment, we inevitably touch on the concept of holistic care, a term also emphasized within the guidelines.
Holistic care focuses on the comprehensive improvement of patient well-being. In our last webinar, we delved into the significance of health literacy and the empowerment of patients to initiate meaningful conversations with their healthcare providers. This approach aligns with the use of quality of life questionnaires, enhancing the patient experience and contributing valuable data to the healthcare system.
I’m pleased to highlight an initiative by the National Health Service (NHS) in the UK, which exemplifies good practice in this area. They have implemented an annual review process, mandating healthcare providers to report on their use of quality of life questionnaires. This practice not only bridges the gap between patient and healthcare professional perspectives but also closes the loop in the patient care cycle.
Moving on to the guidelines, as previously inquired by Andreas, we categorize recommendations by color and level of significance. We’ve discussed the green recommendations, including the importance of integrating patient-reported outcomes within the multidisciplinary team. Our last webinar covered the recommendation on patient-centered collaboration with patient associations. In our next segment, we will delve into the red-framed recommendations, focusing on emergency response and lung health.
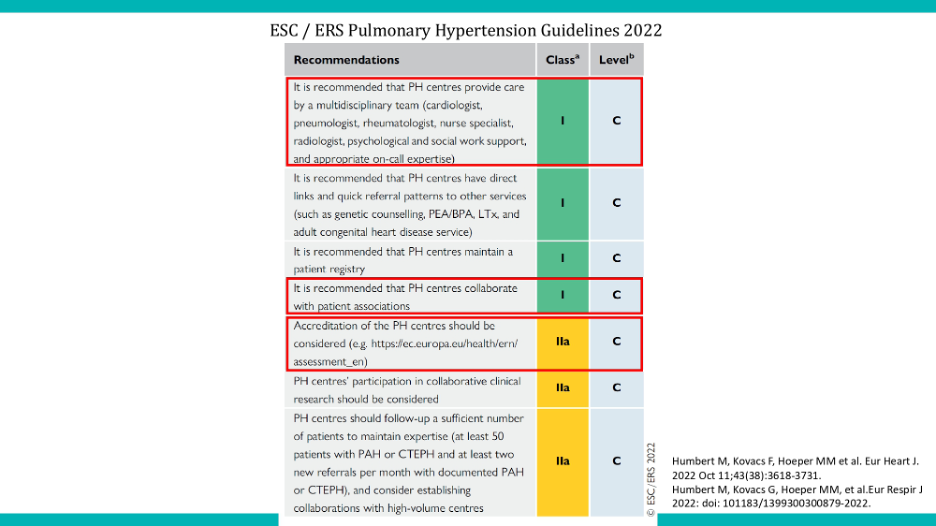
To summarize, we’ve explored the theoretical aspects of quality of life questionnaires and patient-reported outcomes from both the authority’s and guideline’s perspectives. Now, let’s transition to understanding these concepts from a clinical standpoint, examining their application in actual patient care. Simon, over to you.”
DR. SIMON GIBBS
Thank you very much, Gergely, for your insightful contribution. The concept you introduced, that patient-reported outcomes in pulmonary hypertension currently exist in an ‘evidence-free zone,’ is particularly intriguing. It underscores a global perspective that integrating patient perspectives into clinical care enhances the quality and effectiveness of that care. This has been demonstrated across various diseases and in many countries. However, it’s important to note that such evidence primarily comes from the Global North, with limited data from the Global South.
In clinical care, the question arises: How significant are patient-reported outcomes in pulmonary hypertension? Let’s delve a bit deeper into their role. Traditionally, clinical observation, patient discussions, examinations, and investigations have been the primary methods for assessing a patient’s condition. However, these methods fall short in fully capturing the quality of life from the patient’s perspective. There is often a significant discrepancy between physician and patient assessments of quality of life, highlighting a gap in understanding and empathy.
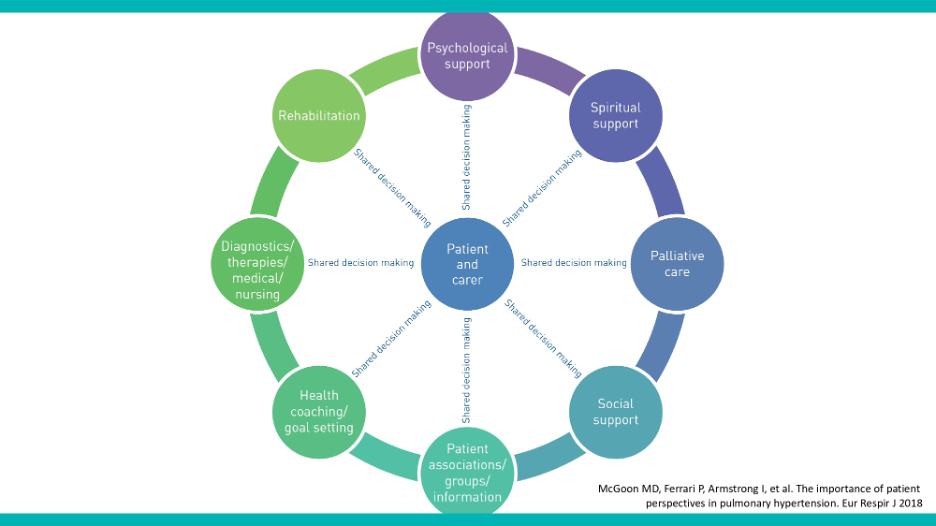
Patient-reported outcome measures offer a systematic, comprehensive, and robust way to bridge this gap. They not only complement clinical assessments but also provide invaluable insights into the patient’s needs and experiences. They can significantly impact patient management, offering a broader scope for discussion and shared decision-making, which was the focus of our last webinar.
Patient-reported outcome measures excel in identifying psychological or psychiatric risks, such as depression, and facilitate discussions on a wide range of support options, including psychological, social, and financial support. They enable a multidisciplinary approach to patient care, ensuring that treatment is individualized and comprehensive.
It’s crucial to recognize that while patient-reported outcome measures have shown to narrow the gap between patient and physician perceptions in other diseases, there’s still a need for data to demonstrate their impact on health outcomes in pulmonary hypertension. The challenge lies in defining and measuring ‘better health,’ a concept that patient-reported outcome measures have struggled to quantify due to the variability in study designs and outcomes.

As we consider the role of patient-reported outcome measures in pulmonary hypertension, we must ask ourselves about their application and effectiveness in this specific context. Gergely, do you utilize them in your practice? How do they influence patient care in pulmonary hypertension? This discussion opens the door to further explore how patient-reported outcome measures can enhance patient-centered care and potentially improve outcomes in this challenging field, what do you say about it Gergely ?”
GERGELY MESZAROS
Thank you, Simon, for providing an excellent foundation for our discussion. As I previously mentioned when we explored the guidelines, there’s a clear need for more evidence and scientific research to support the theoretical basis behind these forms. In the guidelines, we reference several papers specific to pulmonary hypertension (PH), highlighting patient-reported outcome measures as potential independent prognostic indicators for patients.
A noteworthy paper from the UK—where, as I often say to Simon, we have much to learn—further emphasizes the significant impact of pulmonary hypertension on overall health-related quality of life.
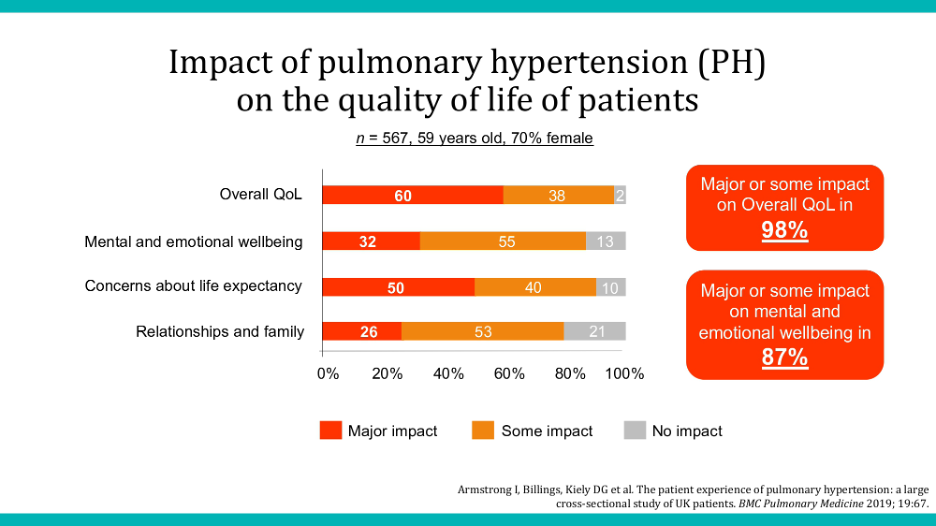
However, without proper evidence, these insights cannot be formally integrated into scientific literature. This underscores the importance of collaborative efforts with physicians on projects like this. From a patient’s perspective, a decline in quality of life might indicate issues with treatment, even if medical records suggest stability. This discrepancy highlights why we advocate for the use of patient-reported outcome measures and patient-reported outcomes, supported by scientific data.
The 2019 paper specifically underlined pulmonary hypertension’s major impact on patients’ quality of life, including well-being and mental and emotional health. The data leave little room for doubt about the disease’s effects, underscoring the need for comprehensive treatment approaches that address more than just clinical symptoms.
Next, I’d like to introduce another initiative under the European Reference Networks (ERNs), specifically the European Reference Networks for Rare Lung Diseases (ERN-Lung). Established in 2017 by the European Commission, European Reference Networks focus on various disease groups, including rare respiratory diseases. ERN-Lung is part of this broader network, facilitating knowledge exchange and patient-centric care. It supports the implementation of cross-border healthcare directives and funds projects that might not attract pharmaceutical interest.

I’m particularly excited that Simon has taken the lead on the patient reported outcome measures functional committee within ERN-Lung. This involvement spans multiple disease areas, such as cystic fibrosis and idiopathic diseases, allowing for a rich exchange of knowledge. Our pioneering efforts on PROMs have led to a project that Simon will detail further.
DR. ANDREAS REIMANN
May I ask a question that might seem simplistic but is prompted by the map we’re looking at? I noticed Sicily, Corsica, and Sardinia seem to be excluded from the European Reference Center Network coverage, despite being integral parts of Italy and France. Could you clarify why this is the case?
GERGELY MESZAROS
Yes, it seems the issue might be related to the map’s coloring or graphical representation. It’s possible that the apparent exclusion of Sicily, Corsica, and Sardinia simply reflects the absence of hospitals from those specific islands participating in the network. However, rest assured that Italy and France, including their island regions, are integral parts of the European Reference Network. Thank you for pointing this out. Pointing this out so it might be some coloring issue.
DR. ANDREAS REIMANN
Absolutely, and the key point to highlight, Simon, which I know you’ll address, is the UK’s continued participation in the European Reference Network despite Brexit. This is an important aspect for our discussions.
DR. SIMON GIBBS
Despite Brexit, the UK remains involved in the European Reference Network, although not as a full member. This technicality, which limits the UK’s voting rights, doesn’t significantly impact the collaboration. From my perspective, the most exciting aspect of the European Reference Network is the extensive patient involvement. Patient organizations and individuals actively participate, making it an ideal platform for working with patient reported outcome measures. This aligns perfectly with our efforts to integrate patient reported outcome measures into our work.
We conducted a survey between the end of 2021 and the beginning of 2022, gathering responses from 30 pulmonary hypertension centers and 37 physicians within those centers.
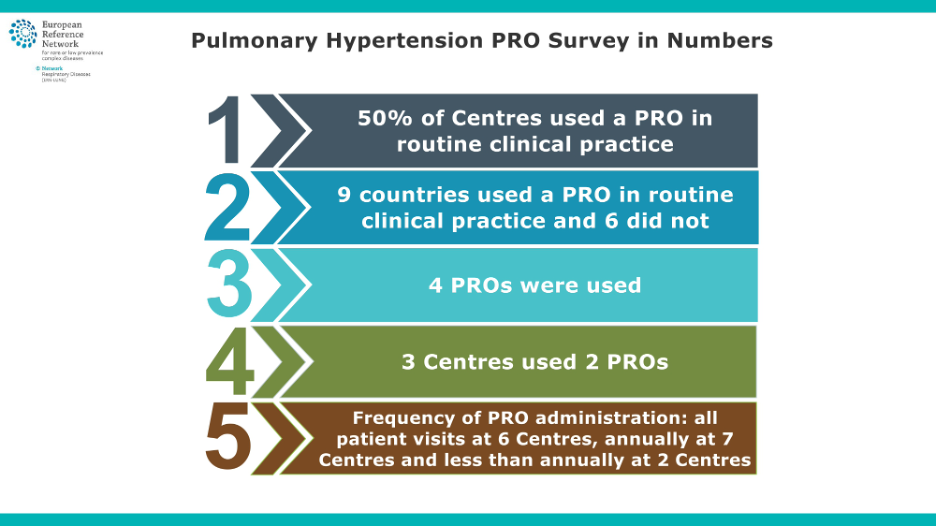
The key findings revealed that, at that time, only half of the centers were using patient reported outcome measures in routine clinical practice. It’s important to note that the other half weren’t necessarily in the wrong; patient reported outcome measures were not a guideline requirement before last year. However, since the survey’s conclusion, the adoption of patient reported outcome measures has increased.
The survey also highlighted that four different patient reported outcome measures were in use, two disease-specific and two generic. The frequency of patient reported outcome measures administration varied widely, from every visit to once a year or even less, reflecting the lack of data on optimal frequency.
I’d like to hear Gergely’s insights on what these findings might imply for pulmonary hypertension care based on this survey.

GERGELY MESZAROS
Thank you, Simon. We were indeed encouraged by the findings of the survey, which offered practical insights into the implementation of the theoretical recommendations outlined in the guidelines. It confirmed our suspicion that there is no standardized clinical practice regarding the use of patient reported outcome measures. The survey revealed concerns among some healthcare providers about the additional workload and time pressures that patient reported outcome measures might introduce to clinical services.
From a patient perspective, we suggest completing these questionnaires during waiting times or arriving with pre-filled forms to minimize the impact on clinical workflow. However, we acknowledge the time constraints faced by clinical staff and the challenges of integrating sometimes difficult-to-measure data into routine care.
The variety of patient reported outcome measures and patient reported outcomes used across different centers can lead to inconsistencies, especially if a patient moves to a new location where different forms are used, potentially rendering years of collected data non-transferable.
One notable finding from the survey was the lack of consensus on how frequently patient reported outcome measures should be administered, with responses varying from every visit to once a year, or only at the initial consultation. This variability underscores the need for clearer guidelines on patient reported outcome measures usage.
As patients, we face the challenge of ensuring that the deeply personal data we share is used meaningfully and not disregarded. This requires a delicate balance, as we understand the additional responsibilities it places on physicians.
The survey highlighted several challenges and questions that need to be addressed to standardize the use of quality of life questionnaires and measures. Simon, it’s now an opportune time for you to introduce some of the patient reported outcome measures that are particularly significant to our discussion, including one that I believe is especially dear to you.
DR. SIMON GIBBS
Thank you, Gergely. Moving forward, I’d like to highlight that presenting a comprehensive list of all PROMs used in pulmonary hypertension would be overwhelming due to its vastness. Therefore, I’ve selected six patient reported outcome measures for discussion: four currently used in ERN-Lung’s clinical settings and two often seen in clinical trials. This selection isn’t to diminish the importance of other patient reported outcome measures but to manage our discussion time effectively.

Notably, “CAMPHOR” and “EmPHasis-10” serve as examples of how patient reported outcome measures can vary significantly. CAMPHOR assesses symptoms, quality of life, and functioning across three domains, whereas EmPHasis-10 focuses solely on the impact of pulmonary hypertension on life quality. This diversity in focus and methodology underscores the need to choose PROMs that align with specific research or clinical objectives.
The timing of symptom assessment is crucial, as demonstrated by PAH-SYMPACT, which differentiates between symptoms experienced over the past 24 hours and impacts over the last seven days. This distinction highlights the importance of the ‘when’ in patient reported outcome measures, affecting how patients report their condition.

Another key point is the relationship between the number of questions in a patient reported outcome measures and patient adherence. Shorter questionnaires generally see higher completion rates among patients. For example, EmPHasis-10 was designed by patients for patients, emphasizing brevity with only ten questions to fit on a single sheet of paper.
Translating and validating patient reported outcome measures in different languages is essential for their accurate use across diverse populations. Copyrights and associated fees can also pose challenges, particularly in Europe where the variety of languages complicates accessibility.
Lastly, there’s a notable gap in patient reported outcome measures designed specifically for children, requiring the use of generic patient reported outcome measures in pediatric cases.
As we conclude, I invite Gergely to share insights on the role of patient reported outcome measures in clinical trials, highlighting the practical implications and potential challenges in their application.”
GERGELY MESZAROS
Thank you, Simon, for the comprehensive overview. Let’s briefly recap the applications of patient reported outcome measures as understanding their broad utility is essential. We’ve discussed their use in audits and clinical practice for patient management. Beyond these, patient reported outcome measures offer valuable self-reflection opportunities for patients, allowing us to monitor our own progress and well-being. Simon highlighted the potential of incorporating quality of life questionnaires in clinical trials, which opens up significant possibilities.
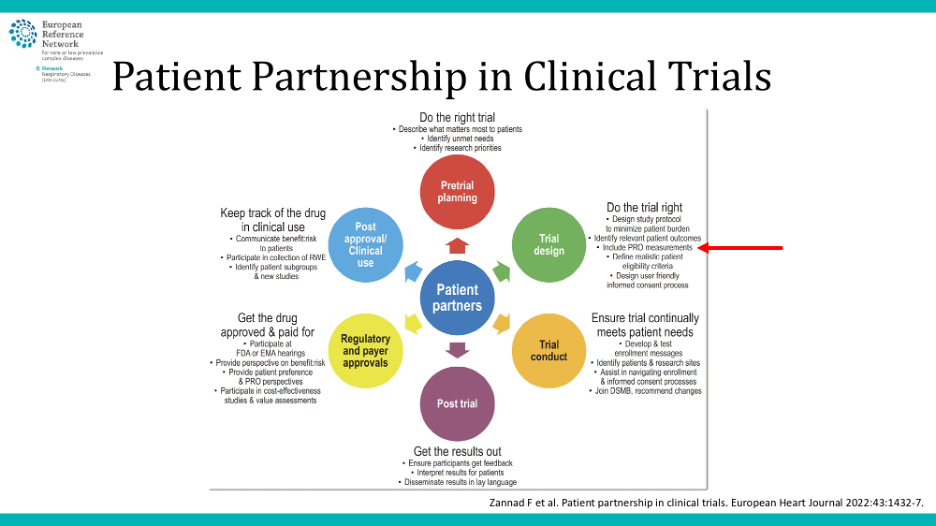
Although we’re pressed for time, I want to stress the importance of placing patients at the center of clinical trials, as indicated in our previous discussions and the recent guideline updates. This patient-centric approach is also evident in trials where patients contribute to the Medical Steering Committee, influencing trial design and endpoint selection. Simon will delve deeper into how patient reported outcome measures and quality of life questionnaires can shape trial endpoints and recommendations.
Furthermore, patients play a crucial role beyond the clinical trial phase, particularly in phase 4 or post-market studies, underscoring our integral role throughout the clinical trial process. This diagram I’ve shared illustrates the systematic inclusion of patient-reported outcome measures in clinical trials, advocating for a step-by-step approach that Simon will expand upon.
DR. SIMON GIBBS
Thank you, Gergely, for your insightful contributions. While we lack the time to delve deeply into the World Symposium for Pulmonary Hypertension (WSPH), it’s important to note that this global gathering on pulmonary hypertension convenes every five years to provide updates and recommendations that bridge the gaps between guideline publications.

Since 2008, three key points have emerged:
1. Use of patient reported outcome measures in Clinical Trials patient reported outcome measures are recommended as secondary endpoints, meaning they complement but do not determine the trial’s outcome. This supports primary endpoints like the six-minute walk test distance in pulmonary arterial hypertension trials.
2. Patient Involvement in Trial Design. The inclusion of patients in designing clinical trials is crucial. Their perspectives ensure that trials address patient-centric concerns, emphasizing the need for expert patient representatives on clinical trial committees.
3. Patient reported outcome measures as Co-primary Endpoints; There’s a proposal for patient reported outcome measures to evolve from secondary to co-primary endpoints in trials, subject to regulatory authority approval, which is essential for drug licensing.
As we wrap up, I hope our discussion has illuminated the significance of patient reported outcome measures, their potential future role, and the importance of patient and caregiver perspectives. To the patients and caregivers in our audience, we’re eager to hear your views on several questions:
Should patient reported outcome measures be routinely used in clinical care for pulmonary hypertension?
How often are you willing to complete a patient reported outcome measure, given today’s busy schedules?
Could patient reported outcome measures serve as a quality measure for clinical services?
Finally, is there a need for a clinical trial to verify if patient reported outcome measures directly improve patient health outcomes in pulmonary hypertension, or are enhancements in communication, patient satisfaction, and quality of life sufficient?
Thank you all for your attention and engagement.
DR. ANDREAS REIMANN
Thank you very much, Simon and Gergely, for your thorough presentation on the use of patient reported outcome measures in pulmonary hypertension care. This discussion raises several important questions, particularly about the routine use of patient reported outcome measures in pulmonary hypertension management from both patient and physician perspectives. The guidelines suggest considering patient reported outcome measures, acknowledging the progress we’ve made, yet also highlighting the need to address concerns raised.
One critical question that has emerged pertains to the balance between the systematic use of patient reported outcome measures for enhancing patient care and the potential burden it places on patients. Patient reported outcome measures require time to complete and could complicate patients’ lives further.
From a patient’s viewpoint, especially considering your representation in the European Reference Network, what is your stance on finding the right balance between leveraging patient reported outcome measures for tailored care and avoiding additional stress for patients? How do we navigate the complexities of integrating patient reported outcome measures into routine care without overwhelming patients?
Following this, we’ll also explore perspectives from the healthcare providers’ side.”
GERGELY MESZAROS
Thank you for the question. We’ve observed a growing preference for making questionnaires accessible online, allowing patients to complete them at their convenience. A few years back, the US Pulmonary Hypertension Association (PHA) conducted a survey to gauge patients’ attitudes toward these online questionnaires, including their preferred frequency of completion. Surprisingly, the most favored frequency was weekly, indicating that patients didn’t find it burdensome to fill out questionnaires on this basis. This preference likely reflects the benefit of regular self-reflection on their achievements and areas for improvement over the week.
The survey results strongly supported the shift to online formats, moving away from traditional paper methods. While there might be concerns about older patients’ ease with digital tools, our findings suggest that the challenge lies more with digital literacy than age. This is an area where patient associations can make a significant difference, offering support and training to ensure all patients feel comfortable using digital platforms for their feedback.
DR. ANDREAS REIMANN
Yeah, thanks very much. So there is room for improvement, but it’s applicable. Simon, what do you think?
DR. SIMON GIBBS
Implementing questionnaires on mobile phones can significantly enhance participation, as it offers a quick and easy method for most people. It’s crucial to keep these questionnaires concise and ensure that healthcare professionals engage in discussions with patients about their responses. This dialogue should explore potential areas of support, demonstrating to patients the tangible benefits of their input. Such interactions affirm that their feedback is not merely a formality but a valuable component of their care.”
DR. ANDREAS REIMANN
Indeed, we have time for a few more questions. One inquiry of interest is whether the impact of therapy on a patient’s life is assessed through these patient reported outcome measures, and, importantly, whether this assessment is easily interpretable.
DR. SIMON GIBBS
Passing the question to Gergely, and yes, the answer is definitively yes. Patient reported outcome measures are designed to capture the comprehensive impact of various factors on a patient’s life, serving as an overarching measure of well-being. They assess overall quality of life through patient responses, which can reflect improvements due to medication, lifestyle changes, or other external factors. The goal is to monitor the cumulative effect of all these elements. If there’s a change in the patient’s condition and the only variable is medication, then it’s reasonable to attribute the observed impact to the treatment.
GERGELY MESZAROS
Yes, I’d like to add to that. Apologies for interrupting, but it’s worth mentioning that there is a scientific paper supporting Simon’s points.
There is a documented correlation between Patient reported outcome measures and patients’ overall well-being, including hemodynamic and other functional outcomes. This correlation helps track whether there is progress or not in a patient’s condition. While I can’t remember if these studies were included in the guidelines, there are definitely scientific papers supporting this relationship.
DR. SIMON GIBBS
And some of the pulmonary hypertension problems predict survival.
DR: ANDREAS REIMANN
It’s fascinating to consider. Tessa Lush [from Goassamer Bio, in the audience] has complimented the webinar and raised an interesting point about the potential of mobile apps for daily well-being and symptom tracking, specifically mentioning health related quality of life (HRQL). She referred to an app called SimTrack, utilized for mass patient data collection before healthcare appointments. This allows doctors or nurses to review patient-reported data ahead of consultations, enhancing discussions and providing insights into patients’ good and not-so-good days. This ties back to our previous discussion about the utility of apps for health monitoring.
Simon, drawing from your experience and that of your colleagues, how receptive are healthcare professionals to analyzing data reported by patients through such apps? How prepared are doctors to integrate this patient-produced and documented information into their clinical assessments?”
DR. SIMON GIBBS
There are two key aspects to consider. First, when patients complete the questionnaire just before their appointment, it enables doctors to directly address the reported information during the consultation, enhancing the relevance and immediacy of the discussion. The second point hinges on how the data is presented. The optimal scenario involves presenting the data not just to individual doctors but to the entire multidisciplinary team, including doctors, nurses, social workers, and other professionals involved in patient care. When the whole team has access to this comprehensive view of the patient’s reported well-being, it becomes an invaluable tool during patient discussions, ensuring that the team can make informed decisions and provide tailored support. Thus, the willingness to utilize this data effectively is contingent on its integration into the team’s review process.”
GERGELY MESZAROS
What we’ve gathered from numerous healthcare providers is the current lack of capability to incorporate feedback from quality of life questionnaires into patient records. This limitation extends to the minimum data set being developed for registries, posing challenges. However, I’m optimistic that future adjustments will allow for the inclusion of such valuable information. Thank you.
DR. ANDREAS REIMANN
Thank you all for joining us today, whether this afternoon or evening. A special thanks to Dr. Simon Gibbs and Gergely Meszaros for their enlightening presentation and valuable insights. This webinar has been recorded and will be accessible on the Alliance for Pulmonary Hypertension website. We invite you to join us for our upcoming webinars. Thank you once again, and we wish you a wonderful evening.”
Warmest thanks to
- The speakers Dr. Simon Gibbs, National Heart & Lung Institute, Imperial College, London, UK and Gergely Meszaros
- The patient engagement experts admedicum for the organisational and technical support
- To Dr. Andreas Reimann, Managing Partner of admedicum, for moderating the discussions.

