Download Dr. Weatherald and Dr. McLaughlin’s slides here
The webinar on clinical trials in pulmonary hypertension was hosted by the Alliance for Pulmonary Hypertension on Tuesday, September 17, 2024.
Thanks to our speakers, Dr. Jason Weatherald and Dr. Vallerie McLaughlin for sharing their knowledge in an accessible way for everyone and to Alliance for Pulmonary Hypertension volunteer Alena Adarbehova for sharing her story of participating in two clinical trials.
Video recording of Alena Adarbehova
Transcript “NAVIGATING CLINICAL TRIALS IN PULMONARY ARTERIAL HYPERTENSION”, SEPTEMBER 17, 2024
NB. This transcript can be translated into your preferred language – use orange button at the bottom centre of this page to select it (slides are not translatable).
DISCLAIMER: Despite every effort to ensure the accuracy of this transcript, we strongly encourage all visitors to consult with their healthcare professionals before making any decisions based on the information provided. Additionally, while the quality of Google Translate has improved tremendously in recent years, please remember that it is an automated service and not a human translation.
ANDREAS REIMAN
Hello everybody. I’m Andreas Reimann. I welcome you to our fifth webinar in 2024, the webinar of the Alliance for Pulmonary Hypertension a non-for-profit arganization. A Patient led organization devoted to improve the care and knowledge around pulmonary hypertension. Our today’s topic is really instrumental to progress in pulmonary hypertension and that’s clinical trials. How can patients participate in clinical trials? Why is it important that patients should participate in clinical trials and how are actually clinical trials and pulmonary hypertension navigating clinical trials and pulmonary hypertension is the subject. And I’m very pleased to be joined by a faculty of three extremely knowledgeable speakers. First of all, I welcome Dr. Jason Weatherald. He’s associate professor at the Department of Medicine, lung transplant and pulmonary hypertension programs at the University of Alberta in Canada. He will be joined by Dr. Vallerie Victoria McLaughlin, who is the director of the Pulmonary Arterial Hypertension Program at the University of Michigan, Ann Arbor, US, and both will be sharing their experience and their knowledge on clinical trials in pulmonary hypertension. And then they will be followed by Alena Adarbehova. She’s a pharmacist and a former pulmonary hypertension patient after double lung transplant, living in Abu Dhabi. But she has a history of participation in clinical trials and most importantly, she’s also a volunteer at the Alliance for Pulmonary Hypertension. And before we start, Alena, I’d like to thank you for making this possible together with Pisana Ferrari and other volunteers who organize these webinars and we as at admedicum are happy to support this. So without further ado, I’d like to call on the stage Dr. Weatherald and Dr. McLaughlin for their presentations.

JASON WEATHERALD
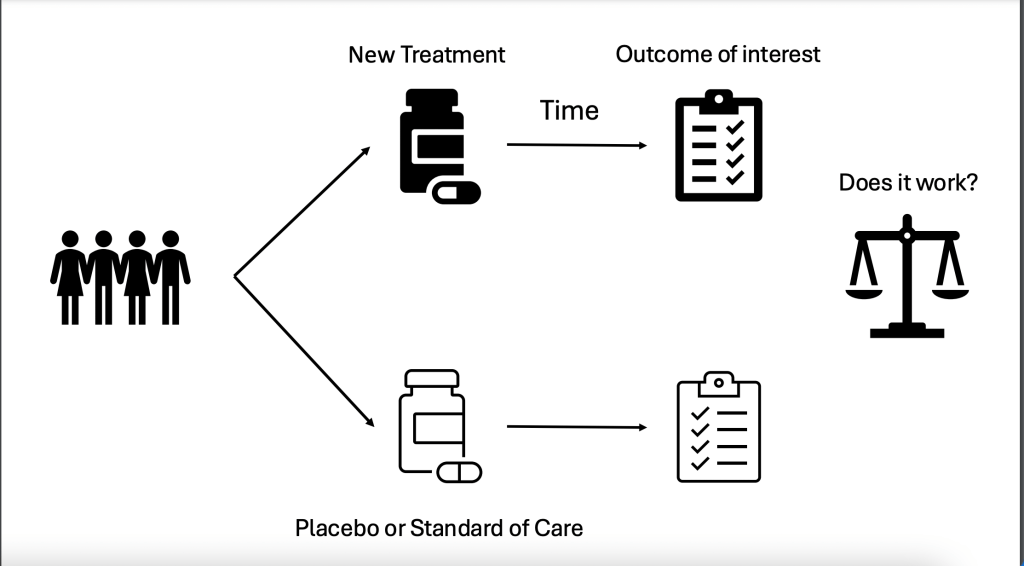
Thank you very much for the introduction. I’m delighted to be today to speak with you all and to share this presentation with you. To start, I just wanted to provide some definitions. So what are clinical trials? Clinical trials are a specific type of research study that involves one or more participants and these people are assigned to one or more interventions of interest. And then we’re looking to see whether or not a specific intervention affects a health related or behavioral outcome. So these interventions could be drugs, vaccines, cells, procedures, radiologic procedures, genetic therapies, natural health products, anything. But in order to show that something works, a clinical trial is required. And at its simplest organization, it involves a population of people with a condition of interest who are then randomly assigned to either the new treatment or intervention of interest or a placebo of that treatment or the standard of care if a placebo is not possible. Then these participants are followed for a certain period of time to see whether or not the intervention actually changes anything. What we’re trying to find out is does it work that is, does it have effectiveness on the condition and is it safe in order to get to market or into the clinic?
Potential interventions have to go through several phases of development or research. This is called the “bench to bedside” pipeline. And this starts in the lab with exploratory research and pre-clinical research using cells and animals to see whether or not a potential intervention might work in people. Once it’s been shown to have some promise in the preclinical phases, it can go into human studies and that goes first of all to phase 1 studies, which are usually in healthy volunteers or in a subgroup of a population of interests such as PAH. If that shows safety and feasibility, then it will go on to a phase 2 study followed by a phase 3 and then ongoing phase 4 research, which is when the treatment is actually in the market and is being used in the clinic. But we’re monitoring for rare side effects or effectiveness in the real world.

So why do we need clinical trials in the first place? As I mentioned before, this is the only way to prove that something is safe, that it’s effective, and it’s really the best way in medicine to advance our knowledge about a treatment and advanced clinical care. We also need them to figure out if there are subgroups of people with a condition that benefit more or if there’s subgroups of people within a disease of interest who do not benefit at all. In order for clinical trial to be meaningful and valid there are a few features that should be present, and this is required not only by the scientific community, but by the regulatory bodies that approve new drugs or devices in the marketplace. First of all, we want a trial to include randomization. This means that participants have a random chance of either being on the active treatment or the placebo or control.
This is the only way to ensure that those groups of people are the same other than the fact that they’re receiving different treatment. This eliminates things called “confounders”. So for example, if we just assigned people to one treatment without randomization, we might find that the age in one group is substantially higher or older than the control group, and it may be that age is one of the factors that explains why a treatment did or didn’t work. So we want to make sure that both groups in a trial are balanced with respect to these potential confounders, such as age or severity of disease.

The other thing we need in a randomized controlled trial is a control. So we need to be comparing the treatment to something. If we just look at one group of people who receive an intervention and then follow them over time, we won’t be able to say what would’ve happened had they received nothing at all. So we need to have a control group, and this might not be a placebo, this could be just the standard of care, but we need to know what would’ve happened without treatment. Ideally, a clinical trial has “blinding”, so this is when the patients and participants in the trial and the researchers conducting the trial don’t know what a participant is receiving. Now, this may or may not be possible to do in certain settings or with certain interventions. It may not be ethically possible to blind, but without blinding, there’s the potential of introducing bias. This means that the patients might behave differently if they know what group they’re in. Similarly, the doctors that are taking care of these people in a clinical trial might do things differently like record the outcomes or treat patients differently if they know what they’re receiving. And this can undermine the validity of a clinical trial.
Also important is that once people are in a clinical trial, that we do everything we can to make sure we have follow-up data on those people. When people leave a clinical trial, this can also undermine the results and introduce bias, especially if the reason for leaving a clinical trial is due to the treatment itself. It may make a treatment look better or worse than it actually is if a large proportion of people in a trial don’t complete the trial.
So what about in pulmonary hypertension? What are we measuring in these clinical trials to know that the treatments that we have work? And I would just like to say that the only reason why we have any treatments today is because of people in the past who have participated in clinical trials that have allowed us to demonstrate that pulmonary hypertension treatments work. So first of all, we have the phase one trials, and this is generally we’re looking at safety and tolerability in a small number of people in phase two trials.
In phase 2 trials we’re also looking at safety and tolerability, but we’re also looking at some evidence that maybe the drug or treatment of interest affects the disease. Typically what we’re doing in pulmonary hypertension phase two trials is hemodynamics. Those are the numbers that we get from a right heart catheterization. We want to know does a drug or a device actually affect the pressure in the lungs or does it affect the function of the right side of the heart? If a treatment is shown to be promising in a phase 2 trial, then we would go on to a phase 3 trial. This is the big trial that shows whether or not a treatment will be approved by regulatory bodies and based on the United States FDA requirements. A phase 3 trial has to have an endpoint. It has to be measuring something that reflects how patients feel function or survive. And what do we do in a clinical trial to measure those?

Well, we might measure symptoms like the functional class on a scale of one to four. We may apply surveys like patient reported outcomes (PROMs) such as the Emphasis 10 or the PAH impact questionnaire. And we also want to measure how does a patient function, what can they do? And the best most standardized way of doing that is the six minute walking distance. A treatment that’s improving the disease should in most cases, improve the ability of a patient to walk farther in six minutes. In larger trials, we may actually look at clinical outcomes, which are things like hospitalizations or clinical worsening, also survival, and importantly, we’re still really concerned about safety and tolerability of a treatment. I’ll pass it over to Dr. McLaughlin now.
VALLERIE McLAUGHLIN
Great, thanks Dr. Weatherald. Well, I’m going to kind of go into a little bit more detail and talk about some of the relevant things specifically for pulmonary arterial hypertension. And we’ve reviewed this a little bit already. So the whole point of going through these trials is so that investigational agents could be approved by the regulatory authority. So drug development is a process that starts in the lab, often goes through animal testing in terms of regulatory issues. Once there’s a new drug, there needs to be something called an IND application. So that’s in the US to the FDA. And I’m sure there’s something similar in the EMA and it’s only after the review of the animal testing and safety that an IND is issued, so that trials can start in humans. Jason reviewed that phase 1 tends to be in healthy patients to just try to get an understanding of dose and side effects.
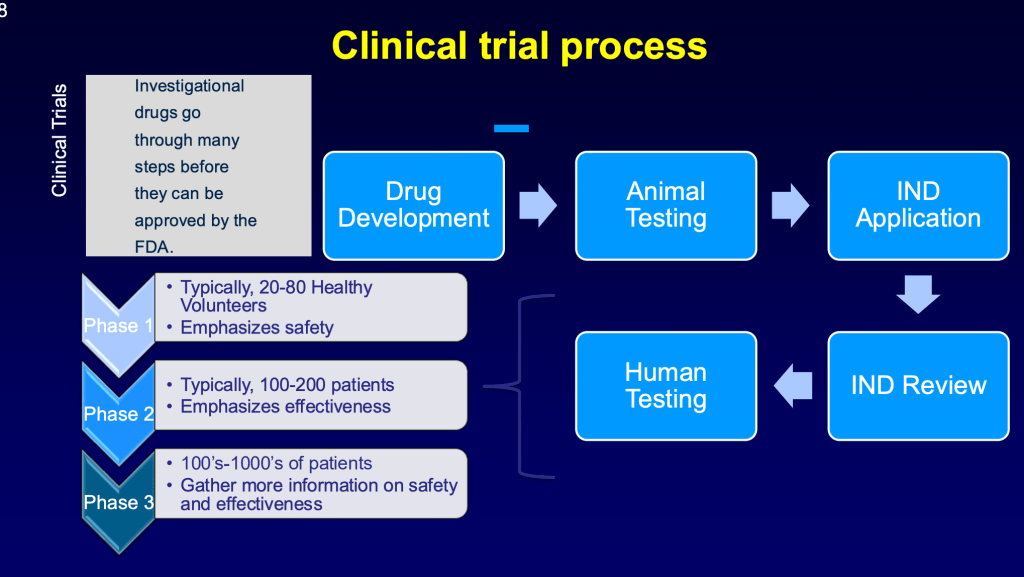
The phase 2 where he mentioned that the right heart cath numbers, the pulmonary vascular resistance is typically the most commonly used endpoint for patients. Those are often much more intense trials with much more testing because we’re trying to learn as much as we can about the drug to make a decision about whether to go into phase 3, so often many more frequent visits. Another important goal of phase 2 is to figure out what is the right dose for further testing. And then phase 3 trials is to gather more information on both safety and how effective the drug is. And those endpoints can range and that can also influence the duration of the trial.
Now I want to go into a little bit more about the regulatory process. This is a part that sometimes patients don’t see. So once the IND is issued, once the sponsor and that sponsor is usually a pharmaceutical company, sometimes a device company in pulmonary hypertension, the sponsor develops a protocol for either the phase 2 or phase 3 trial. Often they gather advice from sometimes a steering committee, sometimes they call it an advisory committee, people like Jason and I who are in this pulmonary hypertension world and have done trials before and can help advise them on things that work, things that don’t work, things that they need to pay attention to. But even with those advisors, they still need to be reviewed with the regulatory agencies because the sponsor wants to know that the regulatory body, be it the FDA or the EMA is in agreement with the protocol, would accept the results as something that would lead to data that would enable a drug approval.
Once they have that protocol and they vetted it through the regulatory agencies, they choose sites for participation. And many of our trials now are multinational trials. So they’re done in many different countries, which has a whole other layer of complexity because right right now most medications are studied on top of background medicine and the background medicine is different in one country versus another. There are some countries that have access to everything but there are some countries which have very limited access to maybe just one or two medications. So you need to consider that in, for example, the randomization process that Jason talked about. You want the patients, it looked pretty similar with respect, not just to age, which was the example that he used, but to the background therapy of pulmonary hypertension. So it’s really important to think about the different sites. And often these are companies called contract research organizations that are used to help the sponsor do this.

Then there’s a whole process of what we call “institutional review board”, and that’s a process that is really in place to ensure patient safety to ensure your safety as you participate in a clinical trial. Some sites use a central IRB, so that’s kind of a specific company that reviews the protocol, reviews the informed consent. Some institutions don’t allow a central IRB and their own institution has a committee of doctors and other researchers and administrators that reviews the protocol and really performs checks and balances to make sure that patient safety and informed consent is being insured.
This is a part that probably many patients don’t think about – the contracts. This is a legal contract between a sponsor, a CRO and an institution, and everyone needs to protect those things and confidentiality needs to be protected. And then there’s budget negotiations as well. And I want to clarify that sites get paid to do clinical research, but they really get paid to do the study related procedures and for the time of the individual. So you need a right heart cath at the beginning and end of the study. That doesn’t go to your insurance. The sponsor is paying for it, so that needs to be negotiated. We have clinical trial coordinators that spend time with you and arrange your visits and administer the PROs. They have salaries that need to be paid. So the budget negotiation, it’s not so that any one site can make money off of a trial, but it’s so that they can pay for the studies and the personnel that are involved in the trial.
The informed consent process is really important. It’s one of the tenets of clinical research, and it’s very important that the informed consents are written in lay language so that the patients can understand it and that they have the opportunity to ask questions. Many of these documents are 10, 12, 14 pages long because it really goes through every potential side effect, every potential risk, every procedure, every study visit. And so it’s important that the patient really thoroughly consider that because we want patients in trials who are going to stay in the trial so that there’s useful information. There’s no point of putting a patient in a trial when they know they’re going to Florida for the winter and they’re not going to be there for the second half of the study. So we want to make sure that patients understand that. And at our site, we take that really, really seriously and we will go through the informed consent with the patient, but we’ll ask them to take it home and read it in more detail and we won’t even get their informed consent until they’ve had at least a week or so to read the document in detail and discuss it with their family. It’s really an important issue. And then there’s training at each site, so it’s not just the doctors and the study coordinators. There’s lots of people who need to be trained on the specific protocol, so the people who are doing the hall walk or the people who are doing the right heart cath or the pharmacists that are responsible for dispensing the investigational drug. There’s a lot of people involved, and so we want to make sure all those people are following the protocol.

So for patients, what to expect, I think the first thing is to acknowledge that study participation is completely voluntary, and this is part of the informed consent process. I think it’s really important for the study team or the physician taking care of you to talk to you about studies for which you’re eligible, but also talk to you about all of the commercial care that you can get. And sometime it’s better for you to go on a commercial drug rather than go into a study which is placebo controlled in which you might receive placebo for a period of time. It depends on your individual situation, and it’s really incumbent upon your provider, your doctor, your nurse practitioner to give you all of the options and not try to push you into a study. The study visits can sometimes be very frequent, and in fact, sometimes really onerous, especially in a phase 2 study, you might need to be there once a month. You might need to have a right heart cath at the beginning and end. So oftentimes the sponsors will help pay for some of that burden. So if you need to stay overnight because you have a bunch of visits one day and a whole bunch of visits and test the next day, they may pay for your hotel stay overnight or pay for your mileage reimbursement or give you some sort of a stipend to buy meals.
On the days that you have a lot of tests, there are a lot of medical assessments, and this is all listed out in the informed consent, tells you what studies are done, at what points in time, so many studies that you might be familiar with such as ECHO or right heart cath. But a lot of studies are looking at other things like patient related outcomes or patient reported outcomes. So there are questionnaires that maybe you haven’t taken before. So again, that is all outlined in the informed consent and the study related assessments are paid for. None of that goes to insurance, but it’s also important to know that should you bring up a different symptom, a new cough at a study visit and an X-ray needs to get done, then that is really more clinical standard care that might go to your insurance. So the study related things should go to the study.
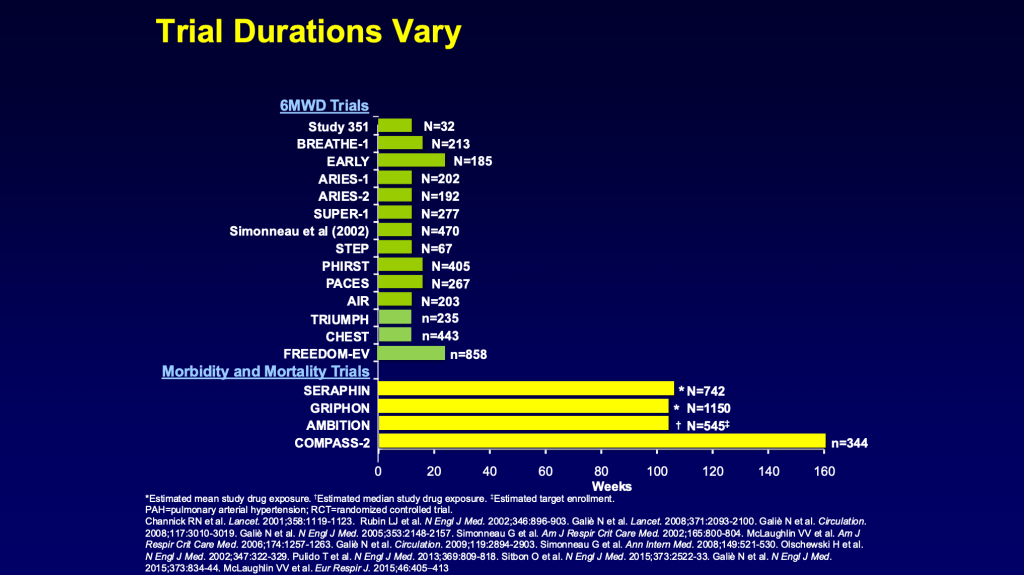
So one of the questions is how long am I in a study? And that really depends on the study and this slide, it’s a little complex, but it tells you about some of the different types of studies that we have. And early on, the earlier studies are at the top. Early on we did a lot of studies that were 12 weeks or 16 weeks and looked at something like six minute hall walk as a primary endpoint. And then we had a number of drugs available and we kind of progressed and we said, shouldn’t we expect more than a six minute hall walk? And we started to move in the direction of some of the other big heart failure trials where we said, it’s not just a hall walk, it’s what happens to the patients, how the patients do over time if they get hospitalized, if they need a lung transplant, if they need to go on more complex therapy.
And there was this push towards what we call morbidity and mortality trials. And these trials are what we call endpoint driven trials. And so the trial lasts until enough patients have been enrolled and have had an event, be that a hospitalization, or a need to go on a pump, or a worsening pulmonary arterial hypertension definition or a death. So any one of those endpoints, and no one knows how long it’s going to take to accumulate the number of endpoints required to stop the trial and analyze it. And so these trials, as you can see, have been much longer, even years for these trials. And that’s important to a patient because as they come in a trial, they need to understand some trials are, you’re potentially on this placebo for 12 weeks or 16 weeks, but for endpoint driven trials, you could be on the placebo for a number of years.

Now there’s pluses and minuses to these different types of trial, and we always have to think about what’s relevant to the patient, what is a meaningful outcome to the patient. And I think we’re starting to get away from the morbidity mortality type trials. Jason may talk about that a little bit more later, but the point is that the trial durations can vary depending on the design of the trial. So let’s talk about the benefits and risk of a trial to a patient. And I’ve listed some here. There may be others that Jason wants to add, but the potential benefits are it’s closer monitoring of your symptoms. You’re seeing much more frequently and you’re tested a little bit more frequently. Sometimes you might get an earlier access to an effective therapy that causes clinical improvement and you may get better sooner. I think it’s always empowering for patients, and I can’t wait to hear from our patient today to play a more active role in your care.
And then there’s also the greater good. As Jason said earlier, we wouldn’t have any of the therapies available for pulmonary hypertension today had it not been for patients who have gone into clinical trials in the past. And some patients are actually doing okay, but they say, I want to pay it forward, I want to help. And it’s this sense of contributing to the greater good in helping the pulmonary hypertension community. The potential risks are there’s side effects. These are investigational drugs. They’ve been studied in animals and maybe some healthy volunteers, but by the time they get to phase 2 and even phase 3 trials, they’ve been studied in a relatively limited number of patients with pulnonary arterial hypertension. And so we always learn about side effects during the course of the trial. Some of them are minimal, some of them are more serious. Many of these drugs have reproductive risks. And in the clinical trials that led up to the phase 1 trials tend to be short. The animal trials tend to be short. So sometimes we’re still learning about reproductive risks. Your symptoms may worsen, they may not improve. You may be on placebo, there may be a delay in other care. So that’s a potential risk and something that really is part of the informed consent process and that you need to talk to your doctor or your provider about early on.
There’s always the risk of lack of confidentiality or privacy. We do everything we can to de-identify, ensure this, but the cybersecurity business is taking off, right? There’s hacks all the time and there’s no way to protect against all of this. So that’s a potential risk. And then there are other unforeseen risks. These are medicines that we’re just starting to learn about. So those are some of the potential benefits and risks.
What happens as the trial goes on? So let’s say that all the patients have been enrolled and we’re at that 16 weeks or we’ve had that last event. The data needs to be queried and cleaned. So when the research coordinators put the data in, usually it’s electronic, sometimes they put a decimal point in the wrong place, and there are checks and balances to go through all the data. Sometimes patients have side effects or what we call “adverse events”. And there are what we call queries from the company to learn more about that, to understand it better, to help determine if it is associated with the drug or if they were COVID positive and their cough is from that. So a lot of those things need to be clarified and cleaned up, and then the database is locked and the statisticians analyze it and the results become available.
The phase 2 study is to really learn about does this drug affect the disease? Again, primary endpoint is most commonly that right heart cath number of pulmonary vascular resistance. Does it have effects on other things that are measured, how walk or ECHO or what have you? And is this impacting the disease enough that we think this could be an effective drug and we need to move on to a phase 3 trial, and what is the right dose for that? So phase 2 is generally that learning phase that helps us make what we call that go or no, no-go decision on further drug development. Phase 3, that larger trial that feels function survives endpoint is generally for regulatory submission to the health authorities. And so sometimes it can take six after a phase 3, it can take six to 12 months to put together that regulatory package because the regulatory agencies want to know everything about that trial, every endpoint, they want to know about every patient, they want to know about every side effect. And so it often takes the companies up to a year to put that dossier together to submit to the regulatory agencies.
Once they submit to the agencies, they also have a lot of time for review, both in the EMA and FDA. There’s things that we call “priority review”. Those are usually really breakthrough drugs or first-in-class drugs get a priority review, which is six months for the FDA and closer to 150 days for the EMA, things that don’t get a priority review. So the third endothelium receptor antagonist may not seem like a priority because there’s two others already available that is 10 months for a standard review with the FDA and up to 210 days for the EMEA. And then once the regulatory agency is approved, that doesn’t mean the drug is available tomorrow. So in the US certainly this needs to get on insurance companies formularies. And in the EMA, even once a drug is approved, then there’s price negotiations with each specific country. And so sometimes the drugs are available right away. For example, in Germany, other times the drugs, it can take a long time to go through price negotiations with the country.

My last slide is how do you find out about clinical trials? Well, there’s lots of ways. I think the best, most reliable source is the pulmonary hypertension care team. So in my center, we have coordinators and they literally screen every single patient that we see in clinic so that we know what trials they’re eligible for. So when I go into a patient’s room after I’m done seeing them, I can say, Hey, we have this trial. You might be eligible for it. Would you like to learn more about it? And so I think the team that takes care of you that’s doing research in pulmonary hypertension is a great source. There’s also clinical trials.gov, which is a website and it’s a little bit onerous, but it does have a lot of information. And then of course, webinars like this, there are a lot of work that patient associations are doing to get the word out. So with that, I will pass it back to Jason.
JASON WEATHERALD
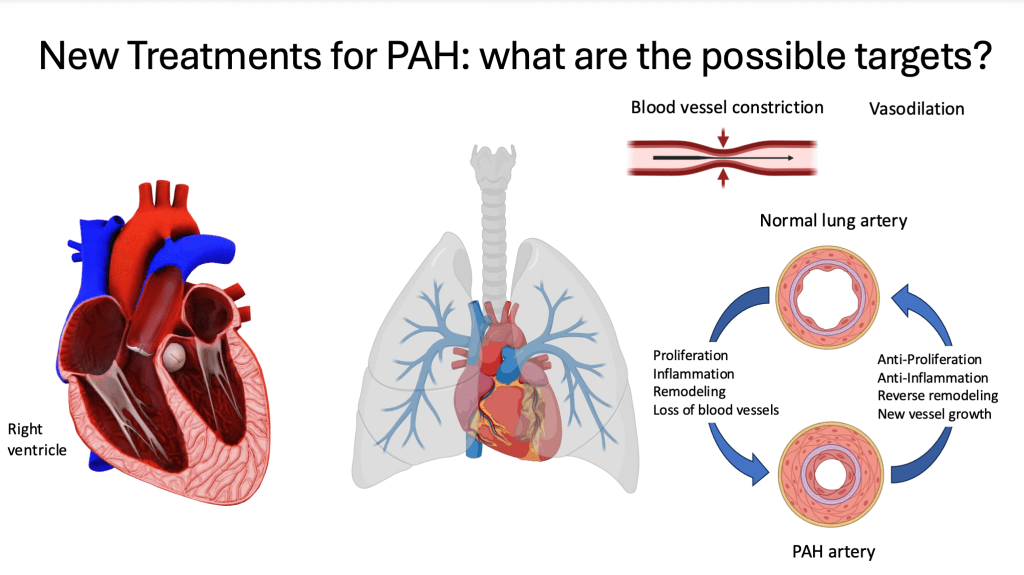
Well thank you for that. And I had like to now talk about some exciting things that are in the pipeline, new treatments that are under development for different types of pulmonary hypertension. So from the scientists point of view, we always want to think about how might we treat this disease better. And so we know that part of the role of the blood vessels in the lungs and pulmonary hypertension are that they constrict, they get narrow. And so we’ve developed drugs that dilate those blood vessels, and this is how most of the available therapies until recently worked. But we know that the blood vessels in pulmonary arterial hypertension are thickened, they’re also enlarged. They have really narrow spaces for blood to flow. And this is due to processes such as proliferation, which is when cells overgrow or get out of control in terms of their division. There’s also inflammation in what we call remodeling as well as just loss of blood vessels in the lungs through various processes.
And so we can think about mechanisms of this disease and how we might target them, like developing therapies that have antiproliferative effects, anti-inflammation effects, drugs or devices that promote reverse remodeling of these blood vessels. And we could even think about therapies that stimulate new blood vessel growth. Importantly, pulmonary arterial hypertension puts a lot of stress on the right side of the heart. And so another possible target are therapies that directly impact the way that the right side of the heart pumps against that high pressure.
This is a busy slide, but I just wanted to highlight that there are a ton of different molecular targets that are being looked at in this disease. So the future is exciting, but as we’ve reviewed, there are lots of steps and hurdles that we have to get past for a drug or any of these mechanisms to make it into the clinic. People are looking at hormonal pathways, metabolism changes to proteins and what we call epigenetic modifications where we can reverse some of the changes to DNA that are underlying the mechanisms of pulmonary arterial hypertension. We can also attack the nervous system, which is abnormal in the lungs. We can look at the cells and what we call the matrix around the cells of the lungs. And we’ve also started to develop stem cell based therapies that could help promote health of the blood vessels in the lungs. We are looking at existing drugs that target inflammation and other diseases and have investigated whether or not these can be repurposed for pulmonary arterial hypertension because we know inflammation plays a role. And there are emerging therapies that target this, what we call the BPR2 or the TGF beta pathway of which sotatercept is a new drug that targets this abnormal pathway. So next I’m going to talk about sotatercept.

This is a new therapy for pulmonary arterial hypertension that is now approved in the United States as well as in Canada recently, and in Europe, which has not necessarily become available to patients yet, but hopefully will be soon. So the way sotatercept works is it tries to rebalance these abnormal signaling pathways in cells in the blood vessels of the lung. So we know that on the right, this BPR2 pathway is abnormal. This is one of the most common proteins that is abnormal in inheritable or genetic causes of pulmonary arterial hypertension. And then there are these pro proliferative factors, and we’re trying to rebalance the abnormalities in the cells. And so sotatercept does that by scavenging these molecules called activin. These are things that stimulate proliferation of cells that we don’t want. And so by removing them or scavenging them, we aim to restore this balance. And so I have some QR codes here if you want to scan them.
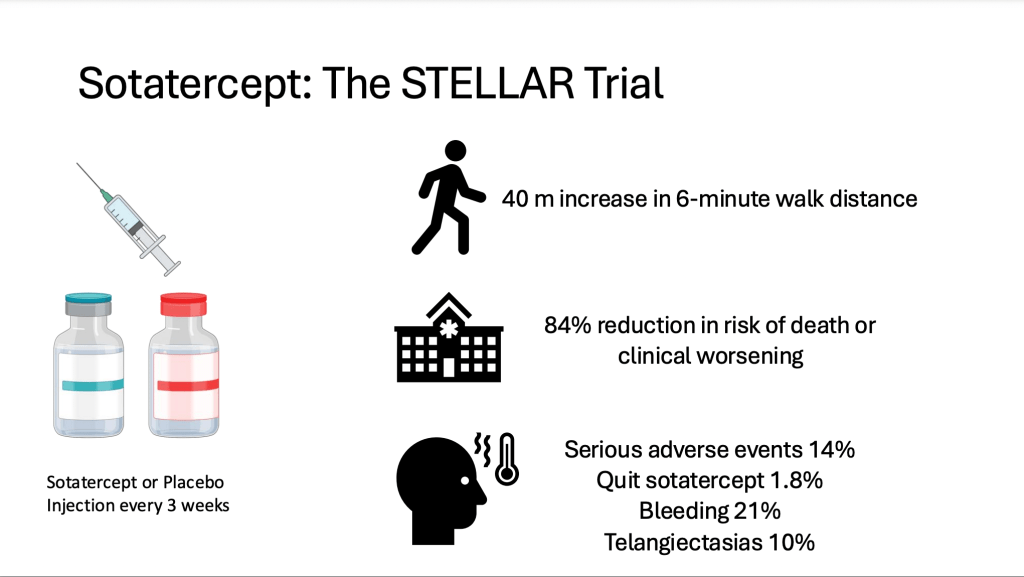
You can go to the actual articles that have been published that have to look at the trials that led to their approval of sotatercept as well as some news releases in the late press such as from CNN. Briefly, how did we know that sotatercept worked? So there was this large clinical trial called the STELLAR trial, and in this trial, patients with pulmonary arterial hypertension, most of whom were on stable background therapy with two or three drugs were randomly assigned to receive sotarcept or a placebo, which is given by an injection in the clinic every three weeks. And the main results of that trial showed that this treatment can increase the six minute walk distance. So that six minute hall walk, which was the main endpoint that was looked at in the trial and on average patients increase their six minute walk distance by 40 meters. And we know from previous studies that that’s clinically meaningful. In general, if a therapy can increase it more than 30 to 33 meters, we consider that a clinically meaningful effect. It also reduced the chance of getting worse or dying during the trial by about 84%. But importantly, there were some serious adverse events or side effects of the drug, some of which led to discontinuation of sotatercept. The most common one that we were seeing in the trial was bleeding, particularly nosebleeds, but also these things called telegiastesia, which are new blood vessels that developed on the skin that kind of look like spiders. And so these have been noted in all of the trials of sotatercept to date. And really the bleeding seems to be the main side effect of concern with this drug that we’ve noticed so far. So it is out there, it’s available in the United States, and as I said, hopefully soon in European countries and in Canada.
But we still don’t know everything about this drug. We know that it worked in a specific population in that phase 3 clinical trial, but we don’t know how long its effects will last. We don’t know if there are rare or long-term side effects with this drug that may emerge years after being on it. The patients in the STELLAR trial were sort of moderate in their severity, and there are other ongoing clinical trials trying to determine whether it works in more severe patients. We don’t know how well it works in patients that have a lot of comorbidities or medical conditions or in newly diagnosed patients. And we also don’t know for sure whether the patients who respond well to sotatercept can safely stop some of their other medications. So these are unanswered questions for the future.
Next, I wanted to talk briefly about this drug called seralutiniib, which is a new potential treatment that has just completed a phase two study with a phase three clinical trial up and running. This is a busy slide, but it just aim to show you what this inhaled molecule can potentially do. It can target many abnormal pathways that underlie pulmonary arterial hypertension. And on the right is just some data from one of the publications showing that seraluitnib when you compare it to no treatment or a healthy control, seem to have effects in animals on the pressure in the lungs, the pulmonary vascular resistance and the NT-proBNP. In this picture in the bottom, it’s just showing some of the abnormal blood vessels in the lungs of animals that were included in that study. So if you wanted to learn more, you can scan this QR to take you to the study. As I mentioned, the phase 2 TORREY trial was recently published, and in this trial, 40 patients were randomly assigned to inhaled seralutinib or a placebo, and it’s administered through an inhaler.
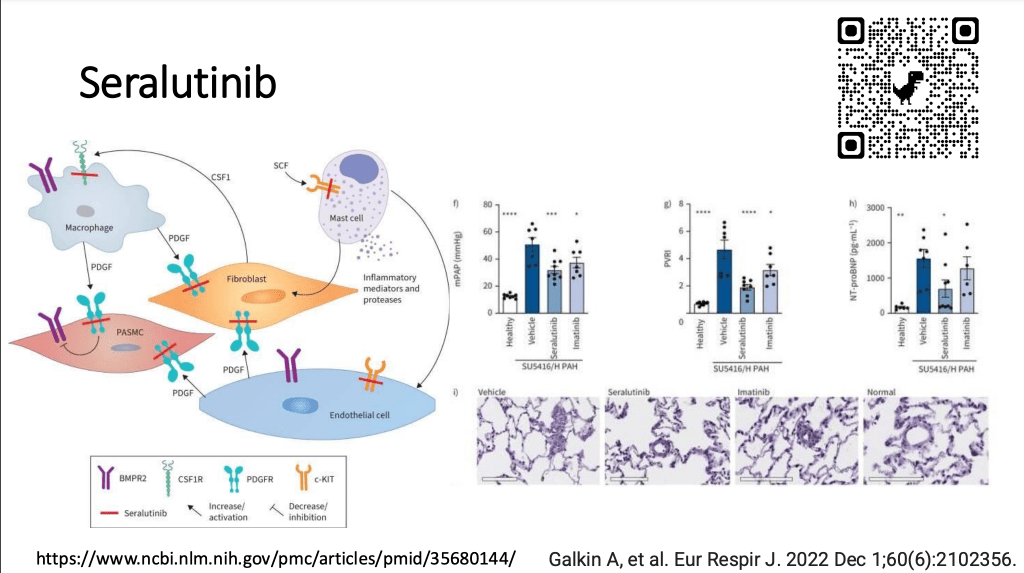

There were 86 patients in total in this study, most of whom were female, and it did improve the pulmonary vascular resistance in the lung. So it dropped that by about 14%, but with a really small potentially not significant improvement in the six minute walk distance. It appeared to be fairly safe in this phase 2 study with cough being the most commonly reported side effect. As I mentioned, the phase 3 trial called PROSERA is now underway. And so this will help us determine whether or not slut actually works in a larger group of patients with a clinical endpoint that is meaningful to patients and to the regulatory bodies.
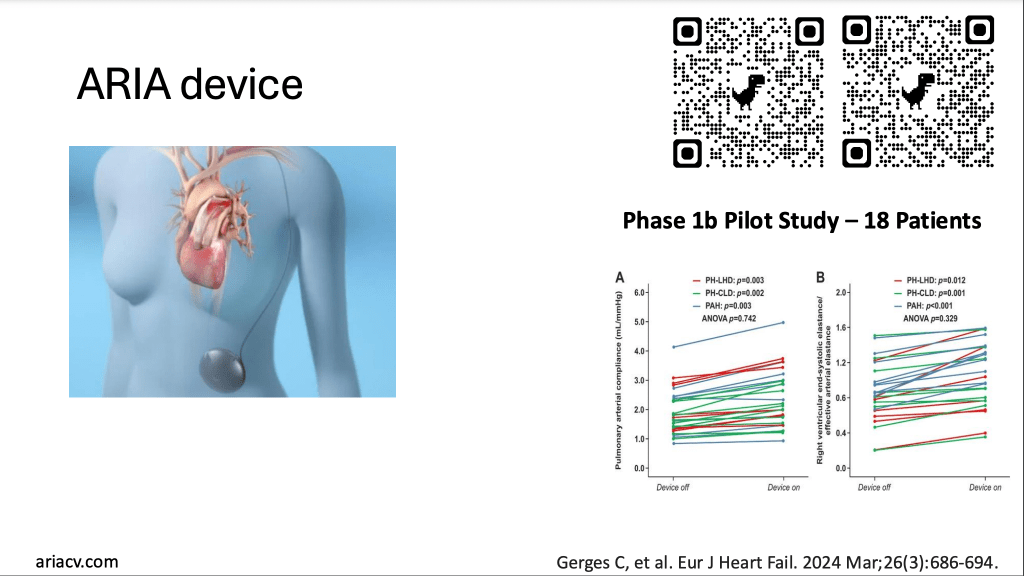
In the last few minutes, I just wanted to look at some more experimental or early phase studies that are kind of exciting. So this device is an implantable device called the ARIA device, and it is an early phase investigation. You can scan this QR code or go to the website to learn more, but effectively it’s a balloon that is implanted in the pulmonary artery of the lung that helps to improve the way blood flow goes into the lung blood vessels and help out the right ventricle do its job. And for the sake of time, I won’t play this whole video, but if you wanted to learn more, you can view this on the website. And this was looked at in a very broad group of patients with pulmonary arterial hypertension in a recent phase 1B study. So a very early phase, not blinded trial that showed that this balloon device did have some improvements in hemodynamics in the lungs. But again, this needs to be investigated fully in a larger group of people.
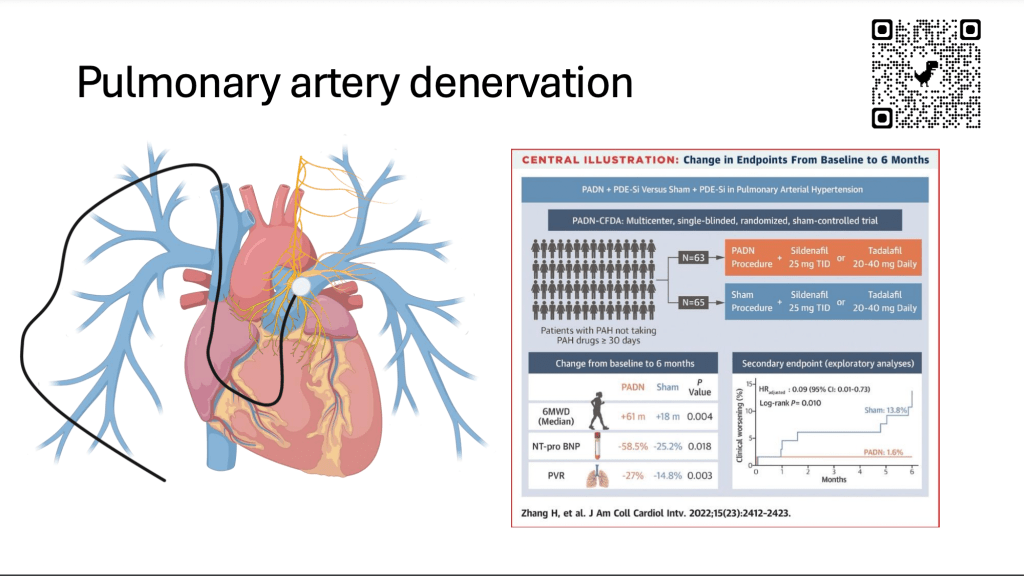
Also, we’ve looked at something called pulmonary artery denervation. This is an intervention, it’s not a drug, but like every cell in the body, the lungs have nerves that go to them. And this comes from the nervous system. And there’s these yellow nerves that follow the blood vessels in the lungs. And we know that these are abnormal in patients with pulmonary arterial hypertension. And so using a right heart catheter approach, this approach can denervate or ablate or sort of zap those nerves so that they stop functioning and stop causing constriction of blood vessels in the lungs. And this has been looked at in some phase 2 and phase 3 trials in different types of pulmonary hypertension. It does seem to have some potential benefits. Importantly, this is probably the largest trial that’s been done so far, but most of these patients were not on background therapy. So a larger trial in sort of a contemporary group of patients who are already treated with best medical care needs to be done in order for this to make it into the mainstream.
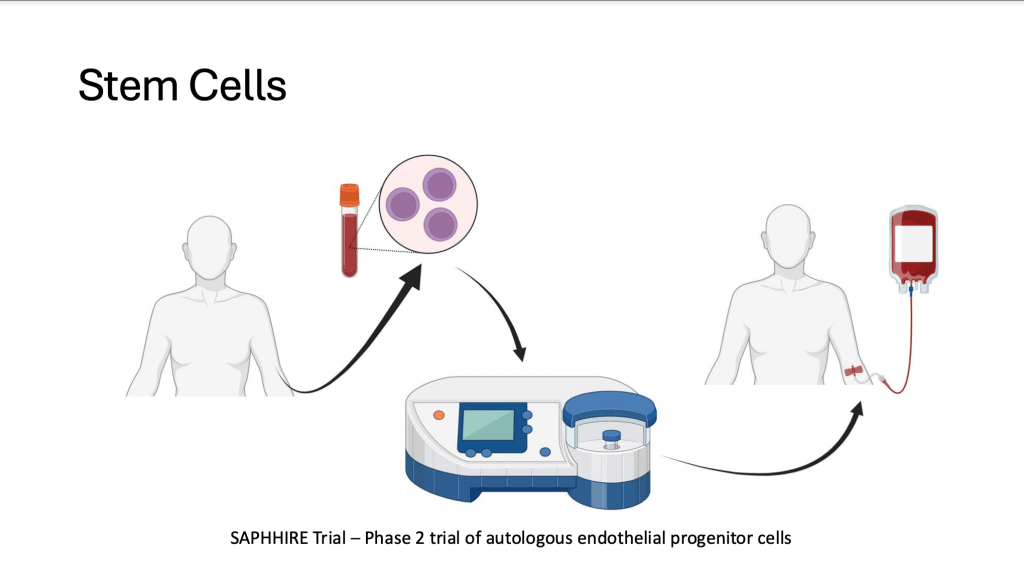
There is a trial that has been completed, it hasn’t been reported yet, called the SAPPHIRE Trial. This is actually a Canadian trial that looked at taking stem cells from a patient, changing them in the lab so that they express more of an enzyme that creates nitric oxide, which is beneficial to the cells in the lung, and then giving patients back their own stem cells. And this was shown in a very early phase pilot study to potentially improve pressure and resistance in the lung. So hopefully we will find out soon whether or not there’s any promise of this approach.
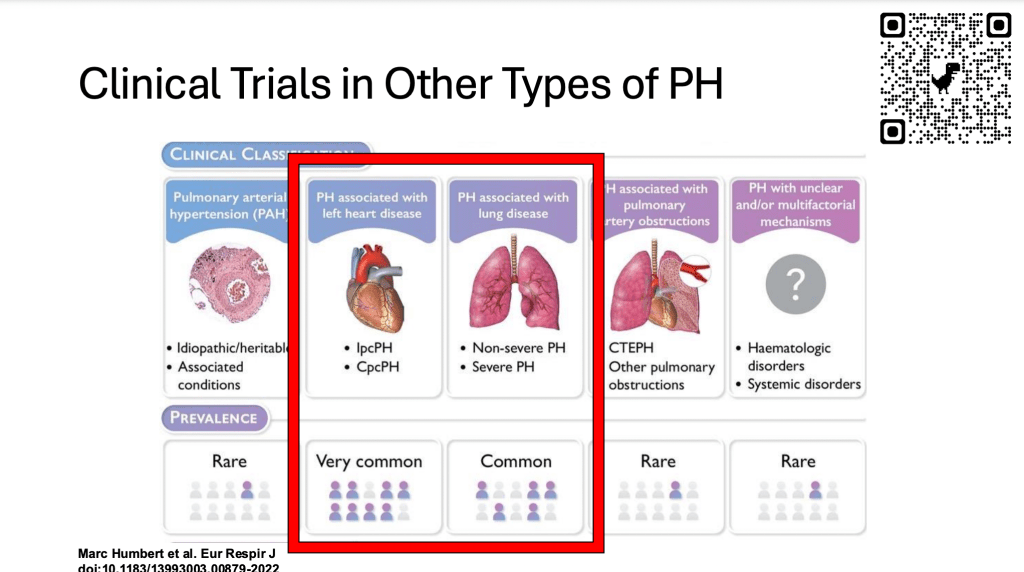
Lastly, I just wanted to talk about other types of pulmonary hypertension. The trials I was just talking about are pulmonary arterial hypertension trials, but there are more common and very important groups of pulmonary hypertension that are due to left heart disease and lung disease for which we really don’t have any good treatments at this time.
And so there are ongoing clinical trials of patients with these more common types of pulmonary hypertension that can be accessed through the centers. And this includes looking at sotatercept, pulmonary artery denervation, other molecules that affect relaxation of blood vessels and also of the heart. And then this drug that has been around for a while called levosimendan in terms of lung disease, pulmonary hypertension. There are other molecules being developed that are inhaled that may improve pulmonary hypertension due to lung disease, as well as a trial ongoing in France looking at tadalafil, one of the pulmonary arterial hypertension drugs in patients with chronic obstructive pulmonary disease (COPD) and severe pulmonary hypertension, so that’s a pulmonary hypertension due to a lung disease called COPD.
So just to summarize, Dr. McLaughlin and I wanted to speak to you about clinical trials and how they’re organized, how they work, and how they need to be designed in order to make sure that they are rigorously conducted so that we know that the results of the trial are valid. Again, this is the only way we can determine whether or not a new therapy really works in patients with pulmonary hypertension. There are a lot of exciting trials ongoing that are aiming to improve upon our existing treatments that target blood vessel dilation, but also to reverse the abnormal proliferation and remodeling in those obstructed blood vessels. There are procedures like denervation that target the nervous system, and there’s also interest in improving the right side of the heart and how it interacts with the lung. With devices like the Aria device and importantly and reassuringly, I think it’s good to know that there are other trials that are targeting those more common types of pulmonary hypertension for which we do not have a lot of effective therapies. And with that, I just wanted to thank everyone for their attention, and we’ll pass it on to the next part of the presentation.

